Therefore, the mass of the water in the container is 2 kilograms.Įxample 3: A swimming pool has a volume of 500 cubic meters. Solution: We are given the volume of water as 2 liters, which is equivalent to 2000 cubic centimeters (2000 cm³).Ĭonverting the volume from cubic centimeters to cubic meters: 2000 cm³ = 0.002 cubic meters (0.002 m³). Calculate the mass of the water in the container, given that the density of water is 1000 kilograms per cubic meter. Therefore, the mass of 500 milliliters of water is 500 grams.Įxample 2: A container has a volume of 2 liters and is filled with water. Substituting the values into the formula: Using the formula for density, Density (ρ) = Mass (m) / Volume (V), we can rearrange the formula to solve for mass: Solution: We are given the volume of water as 500 milliliters, which is equivalent to 500 cubic centimeters (500 cm³). Solved Examples on Density of WaterĮxample 1: Calculate the mass of 500 milliliters of water, given that the density of water is 1 gram per cubic centimeter. Understanding water’s density aids in comprehending natural phenomena, designing structures, and monitoring environmental and industrial processes. Its value of 1 g/cm³ at standard conditions serves as a reference point for the density of other substances. In conclusion, the density of water is a fundamental property with significant implications in various scientific disciplines and practical applications. Physics Formulas Temperature Pressure Conclusion It ensures consistency and adherence to standards during production. Food and Beverage Industry: Density measurements are used in quality control processes to assess the concentration of sugar, alcohol, and other components in beverages.Changes in water density can indicate pollution, mixing patterns, and the availability of nutrients for aquatic life. Environmental Monitoring: The density of water is a key parameter in assessing the health and quality of aquatic ecosystems.It helps engineers determine buoyancy, stability, and the loads that these structures can bear. Engineering and Construction: Understanding the density of water is crucial for designing structures that interact with water, such as dams, ships, and submarines.The knowledge of water’s density finds practical applications in various fields, including: However, this effect is typically negligible compared to temperature and salinity. Pressure: Under high-pressure conditions, such as in the deep ocean, the density of water can increase slightly due to compression.This has implications for oceanography, where variations in salinity can impact water masses and ocean circulation. Generally, an increase in salinity leads to a higher density of water. Salinity: The density of water can be affected by the amount of dissolved salts or minerals in it.This property is the reason why ice floats on water, as it has a lower density than liquid water. This means that warm water is less dense than cold water. 
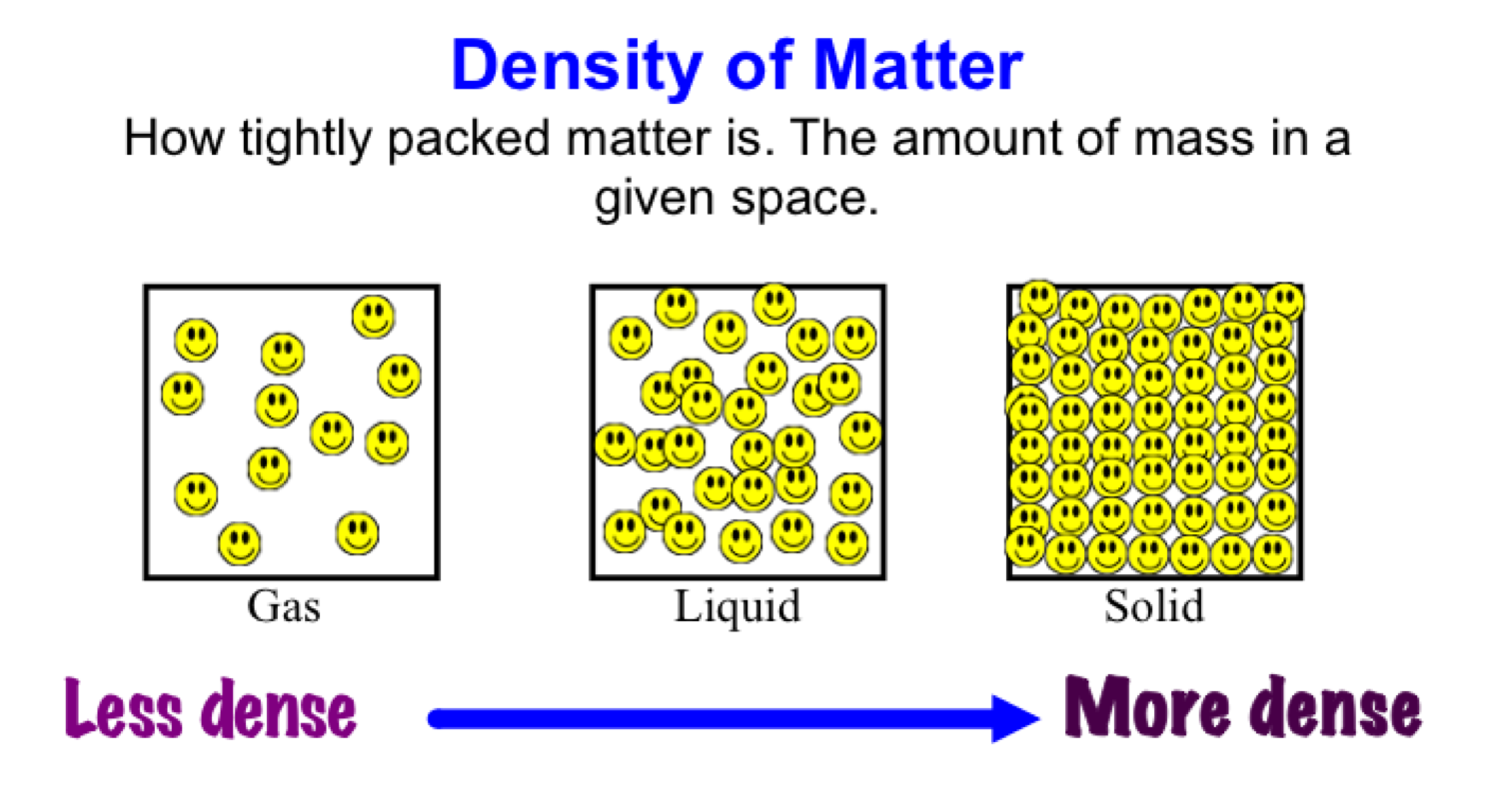
Temperature: The density of water decreases as its temperature increases.The density of water is not constant and can be influenced by several factors: It plays a role in the formation of clouds, precipitation, and the movement of water through different states (solid, liquid, gas) in the atmosphere and on Earth’s surface. Water Cycle: The density of water affects the phase changes of water during the water cycle.These factors impact marine ecosystems, climate patterns, and weather phenomena. Earth’s Hydrosphere: The density of water influences the movement and circulation of ocean currents, as well as the formation of thermoclines, which are layers with varying temperature and density in bodies of water.This principle is used in shipbuilding, designing flotation devices, and understanding the behavior of objects in fluids. Objects that are denser than water will sink, while those that are less dense will float. Buoyancy: The density of water plays a vital role in determining the buoyancy of objects.Here are some key aspects highlighting its significance: The density of water is of immense importance due to its implications in various natural phenomena and human activities. In this note, we will explore the density of water, its significance, factors affecting it, and its applications. Understanding the density of water is fundamental in various scientific disciplines, including chemistry, physics, and engineering. One of its unique properties is its density, which refers to the amount of mass per unit volume. Water is a remarkable substance that plays a crucial role in our lives and the natural world. What is the density of water in CGS unit?.What is the density of water in SI unit?.Why does ice have low density than water?.Why is the water density of water highest at 4o C?.Frequently Asked Questions on Density of Water. 
0 Comments
Leave a Reply. |
Details
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
